CFTR Abundant in Gut, Possible Reason for Intestinal Problems in CF Patients
Written by |
The cystic fibrosis transmembrane conductance regulator (CFTR), a key protein that is defective in people with cystic fibrosis, is abundant in the gastrointestinal tract of people and may be a reason why CF patients are troubled by gastrointestinal problems, a recent collaborative study by researchers at Peking University Health Science Center and Shantou University Medical College, in China, reported.
Findings from the study, “Expression of Cystic Fibrosis Transmembrane Conductance Regulator in Ganglia of Human Gastrointestinal Tract,” were published in the journal Scientific Reports.
Cystic fibrosis (CF) is a genetic condition often characterized by severe pulmonary impairment, but it can also affect organs such as the liver, pancreas, and digestive tract. The disease is caused by a mutant gene encoding the CFTR protein, which plays an important role in the transport of ions, particularly chloride. Problems in this ion channel induces anomalies in fluid transport in cells, ultimately leading to inflammation and fibrosis.
Recent studies detected the expression of CFTR in many parts of the body, including the brain. Now, researchers examined the expression and distribution of CFTR in the human gastrointestinal tract.
The team collected fresh gastrointestinal tissue from 11 surgery patients and five autopsy cases, and analyzed the tissue using various biochemical methods.
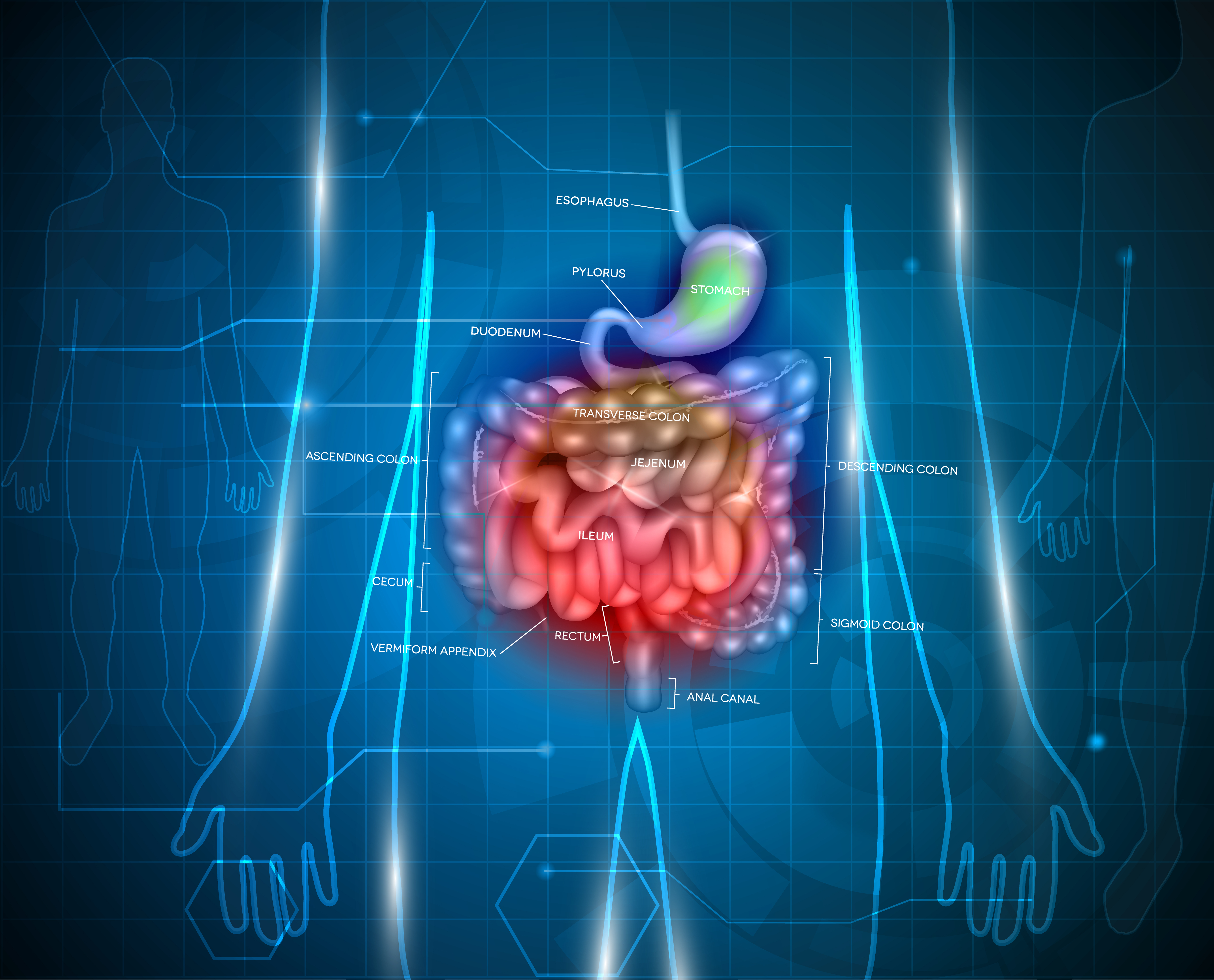
Results revealed the presence of CFTR protein and CFTR messenger RNA (mRNA, an RNA molecule involved in the transfer of genetic information from DNA to proteins) in the gastrointestinal tract, including the stomach, colon, duodenum, jejunum, ileum, cecum, appendix, and rectum. These findings suggest that CFTR may be involved in the physiology of the gastrointestinal tract, which would help to explain why patients with CF often experience gastrointestinal problems.
“In conclusion, our study provides evidence of CFTR expression in the neurons of the enteric nervous system [ENS, the nervous system of the gut]. Widespread distribution of CFTR in enteric ganglia throughout the gastrointestinal tract suggests that this protein might exert a role in maintaining the normal structure and physiological functions of enteric ganglion cells,” the authors wrote.
“In addition, the presence of dysfunctional CFTR in the ENS may have adverse effects on the gastrointestinal tract. Further research is required to clarify the exact function of CFTR in the ENS and the implications of dysfunctional CFTR in the enteric ganglia for patients with CF,” the team concluded.