UK Scientists’ “Molecular Scissors” Technique Could Prevent Genetic Diseases Like Cystic Fibrosis Before Conception
A team of scientists of The University of Bath’s Department of Biology & Biochemistry in Bath, U.K. report that they’ve developed a new gene editing technique that they say will streamline biomedical research and in the future will potentially be harnessed to prevent genetic diseases like Cystic Fibrosis prior to the moment of conception.
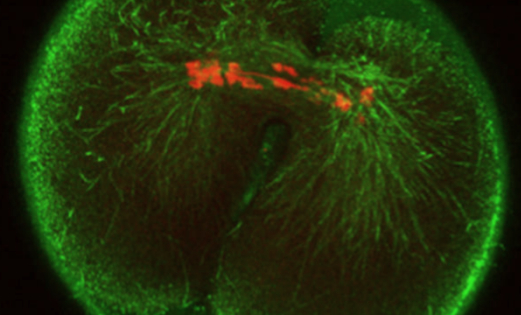
The Bath researchers’ findings have been published in the Nature Group journal Scientific Reports, in an article describing how they have used what they call “molecular scissors” enabling them to edit DNA of either egg or sperm in mice during fertilization, thereby speeding up the genetic research process.
The Scientific Reports paper, entitled “Asymmetric parental genome engineering by Cas9 during mouse meiotic exit“ (Scientific Reports 4, Article number: 7621 doi:10.1038/srep07621), is coauthored by Toru Suzuki, Maki Asami, and Anthony C. F. Perry, all of the Laboratory of Mammalian Molecular Embryology, in the Department of Biology and Biochemistry at the University of Bath
The coauthors note that mammalian genomes can be edited by injecting pronuclear embryos with Cas9 cRNA and guide RNA (gRNA), but that it has been unknown whether editing could also occur during the onset of embryonic development, prior to pronuclear embryogenesis. They explain that gene-targeted mice facilitate functional genetic analysis in vivo, but that the manner in which they are typically produced via embryonic stem (ES) cells is a laborious, time-consuming and expensive process, and that gene targeting in larger species, although increasingly relevant in biomedicine, is even more difficult.
However, the scientists observe that these barriers have recently been negotiated by a powerful newcomer to mammalian genome engineering technology: describes as “the clustered, regularly interspaced, short palindromic repeat (CRISPR)/Cas9 system of adaptable bacterial immunity6, 7. Cas9 is a DNA endonuclease whose site-specificity is determined by a single-stranded RNA.”
The CRISPR)/Cas9 technique was chosen as one of Science Magazine’s Science’s Top 10 Breakthroughs of 2013, referred to as “Genetic Microsurgery For The Masses.”
In their article, the Bath researchers explain how they used the Cas9 enzyme to cut a precise point in the genome, thereby enabling them to accurately inactivate a specific gene report during the initiation of development, noting that maternal alleles underwent editing after a first round of DNA replication, resulting in mosaicism, and that asymmetric editing of maternal and paternal alleles suggests a novel strategy for discriminatory targeting of parental genomes.
[adrotate group=”1″]
They observe that this technique enables scientists to study specified gene functions in mice by creating a “knock-out” in closer to one month rather than the six months typically required when employing conventional techniques, and project that this provides scientists with a powerful tool with which they can substantially accelerate biomedical research and which promises to reduce number of animals required for use in experiments aimed at answering fundamental medical questions.
 Study senior author Dr Tony Perry of the University’s Laboratory of Mammalian Molecular Embryology, explains: “We’re really excited about this research. Previously, this technique had been demonstrated with established embryos but we’ve shown that we can accurately edit genes in the sperm or egg around the time of fertilisation, just as the embryo is starting to develop.”
Study senior author Dr Tony Perry of the University’s Laboratory of Mammalian Molecular Embryology, explains: “We’re really excited about this research. Previously, this technique had been demonstrated with established embryos but we’ve shown that we can accurately edit genes in the sperm or egg around the time of fertilisation, just as the embryo is starting to develop.”
The Cas9 technique works by cutting into the DNA at a precise point in the genome. The cell repairs the damage but “chews” the frayed ends before rejoining them, thereby destroying the gene’s function.
A University of Bath release notes that the technique Dr. Perry and his colleagues have described suggests potential for many exciting potential applications. For example, helping provide disease resistance in livestock or perhaps providing a means of preventing serious genetic disorders in humans at point of conception — for example by allowing carriers of life-threatening genetic conditions like Cystic Fibrosis to conceive healthy babies without risk of passing on the disease.
The researchers also expect that the method could also be used to enable transplant of organs from some large animals into humans, circumventing the problem of rejection, by making the organs “immunologically invisible.”
“This is a dream for transplant surgeons and patients awaiting immunologically matched organs,” Dr.Perry observes. “It means that one day it may be possible to transplant these engineered organs — even if only until a suitable human one is found — and save lives.”
Dr. Perry’s Lab at Bath U. is currently the only research laboratory in the U.K. successfully using piezo-actuated micromanipulation, which is essential for the manipulation of delicate mouse eggs. He notes that all nuclear transfer cloned mice have been made using piezo; and that the lab recently produced England’s first cloned mouse. The scientists can monitor outcomes of piezo-actuated micromanipulation by fluorescence imaging, biochemical analyses and developmental profiling. Dr. Perry says this approach allows his team to harness the biology of fertilization for new technologies, such as a method of transgenesis developed by them, and notes that because egg-to-embryo transition during natural fertilization is ~100% efficient, understanding how it works will prove essential for the development of safe strategies in regenerative medicine.
On the other hand, because it involves genetic manipulation, news of the CRISPR)/Cas9 research success has reignited debate over ethical implications of its application, such as potential for “designer babies.”
A report by BBC News’s health editor James Gallagher cites Dr Perry observing that developments in the field of genetics like precise DNA editing at the moment of conception in mice means that “designer babies” are no longer science fiction. However, Gallagher notes that other leading scientists and bioethicists maintain that serious public debate on associated issues is called for.
As for editing the mouse genome of mice at the precise moment when DNA from sperm and egg become joined, Dr. Perry told the BBC: “We used a pair of molecular scissors and a molecular sat-nav that tells the scissors where to cut. It is approaching 100% efficiency already, it’s a case of ‘you shoot you score,'” and speculating that the CRISPR/Cas9 technique could become a procedure performed during fertility treatment, with the upside being potential for circumvention of heritable disorders caused by DNA mutation such as cystic fibrosis and genetically caused increased risk of cancer, but also cautioning that scientists must proceed with extreme care.
Referencing the BBC article in a January 19 tweet, Dr. Perry says Dr “Society must assess new #geneticengineering technologies & decide what is acceptable.”
The Bath U. researchers have filed a patent application covering their intellectual rights to the technique. Their study was funded by the U.K. Medical Research Council.
Sources:
The University of Bath
Scientific Reports
BBC News
Image Credits:
The University of Bath