Arcturus Presents mRNA Delivery Data Applicable to CF Treatment
Written by |
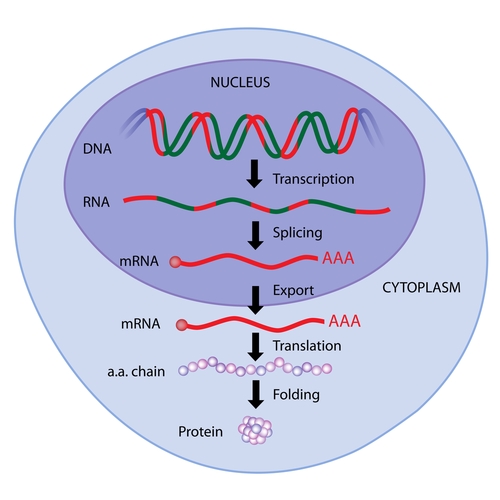
 Arcturus Therapeutics, Inc. recently presented encouraging non-human primate studies concerning its preclinical messenger RNA therapies for the potential treatment of genetic diseases such as cystic fibrosis at the 2nd International mRNA Health Conference held November 11-12 in Cambridge, Massachusetts. Arcturus’ presentation, “Potent Delivery of LUNAR™ Nanoparticles Containing Synthetic mRNA for Therapeutic Protein Expression in Non-Human Primates,” described the safety and efficacy of their novel therapeutic.
Arcturus Therapeutics, Inc. recently presented encouraging non-human primate studies concerning its preclinical messenger RNA therapies for the potential treatment of genetic diseases such as cystic fibrosis at the 2nd International mRNA Health Conference held November 11-12 in Cambridge, Massachusetts. Arcturus’ presentation, “Potent Delivery of LUNAR™ Nanoparticles Containing Synthetic mRNA for Therapeutic Protein Expression in Non-Human Primates,” described the safety and efficacy of their novel therapeutic.
“Messenger RNA (mRNA) offers a powerful way to modulate gene expression within a cell, thereby providing a novel therapeutic approach to treat diseases that were previously inaccessible or relied on expensive and complex protein replacement therapies,” said Dr. Pad Chivukula, CSO and COO of Arcturus, in a news release from the company.
Using mRNA is an attractive option for treating genetic diseases such as cystic fibrosis because instructions for making a proper protein, such as the cystic fibrosis transmembrane conductance receptor (CFTR), are delivered to cells despite the presence of genetic mutations in the cells’ inherent instructions. During in vitro studies, researchers at Arcturus were able to transfect cells with mRNA for CFTR.
[adrotate group=”1″]
Notably, these studies are becoming translatable to humans with the success of LUNAR-delivered mRNA for human erythropoietin. Non-human primates receiving a low dose (0.3 mg/kg) of mRNA saw a 1000-fold increase in erythropoietin protein production with no safety concerns. Further in vivo studies in rodents with alpha-1 antitrypsin, factor IX, and erythropoietin mRNA demonstrated additional promise of the therapy.
“We are thrilled to be presenting our messenger RNA data in rodents and non-human primates, showing impressive production of multiple human therapeutic proteins with a large safety margin due to our proprietary LUNAR deliver technology,” said Dr. Chivukula. “Arcturus is in a unique position having proven successful RNA delivery capability in non-human primates [for messenger RNA].”






